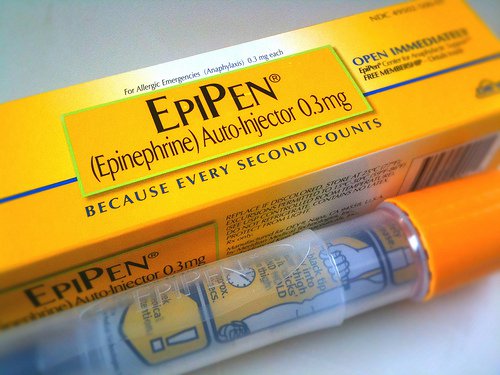
The Food and Drug Administration approved Teva Pharmaceutical Industries Ltd.’s generic version of Mylan’s EpiPen injector device for life-threatening allergic reactions, the most widely sold such device in the U.S., the Wall Street Journal reports.
The product, whose brand-name version is manufactured by Mylan NV, touched off a furor two years ago over its price increases of 548% over about a decade.
The device injects epinephrine into adults and children experiencing anaphylactic and other shock from causes such as bee stings and reactions to food and medicine. Teva, based in Israel, is now authorized to begin marketing its generic auto-injector with epinephrine in two strengths.
The approval of a generic version of this product marks something of a milestone for FDA Commissioner Scott Gottlieb. Dr. Gottlieb has made approval of generics a high priority for the agency, including products commonly referred to as complex generics.
EpiPens are crucial emergency products for people, including many children, who suffer allergic reactions. Mylan was under severe criticism from the public and Congress in 2016 after raising its list price for a pack of two auto-injectors to $609, the Journal reports.











